Elusive Hot Hydrogen Leads To More Pressing Questions - Dr. Ross Howie
MARCH 3, 2015
 Scientific studies of hydrogen at extreme pressures and temperatures are crucial for understanding the rich variety of unusual states and structures, and strange properties, that dense hydrogen demonstrates. But the combination of high pressures with moderate temperatures (>200 GPa and 550–1,300 K) has long been an experimentally inaccessible "no man's land" for hydrogen. Publishing in Nature Materials (http://dx.doi.org/10.1038/nmat4213), Dr. Ross Howie, a new Associate Staff Scientist who is joining HPSTAR in 2015, and co-workers at the University of Edinburgh have demonstrated now to overcome this difficulty, allowing them to observe a new, dense form of hydrogen, possibly a liquid phase at unusually low temperature.
Scientific studies of hydrogen at extreme pressures and temperatures are crucial for understanding the rich variety of unusual states and structures, and strange properties, that dense hydrogen demonstrates. But the combination of high pressures with moderate temperatures (>200 GPa and 550–1,300 K) has long been an experimentally inaccessible "no man's land" for hydrogen. Publishing in Nature Materials (http://dx.doi.org/10.1038/nmat4213), Dr. Ross Howie, a new Associate Staff Scientist who is joining HPSTAR in 2015, and co-workers at the University of Edinburgh have demonstrated now to overcome this difficulty, allowing them to observe a new, dense form of hydrogen, possibly a liquid phase at unusually low temperature.
Recent technological advances have enabled studies of hydrogen under extreme compression and variable temperature, leading to the discovery of no fewer than five solid phases. With its strong quantum effect and extremely high compressibility, the rich physics of hydrogen suggests that other unusual phenomena may occur. A missing piece of the puzzle, however, has been what happens at very high pressure and moderate temperature. This is the key region near the boundary between the fluid and solid state, where new phases and interesting properties are expected, and it has been inaccessible in dynamic studies because of rapid temperature rises during compression. Howie and co-workers have met this challenge by improving the resistively heated diamond-anvil cell technique and hydrogen-containment method, enabling them to reach 570K at 245GPa in pure hydrogen. In this region, they observed a striking change in the optical Raman vibrational spectra, indicating the discovery of a new molecular phase. If this phase is indeed the long sought-after molten hydrogen, as interpreted by the authors, this will be the coldest melt for all known materials above 200GPa. To put this in context, compression usually causes a liquid to freeze into a solid; in very rare cases, such as ice and water, compression causes a solid to melt. Hydrogen would set a record high pressure and record low temperature for pressure-induced melting.
This discovery of a new hydrogen phase represents a significant step forward. It is likely to stimulate the development of key experimental tools and techniques to answer the number of questions its observation raises. For example, is the new phase indeed a liquid? Or is it one of the exotic solids predicted by theory, such as the alternating layers of H2 and graphene-like 3H2 rings? Is the melt conductive? Extension of experimental probes into this region, such as infrared spectroscopy, electrical conductivity measurements and X-ray diffraction, in addition to Raman spectroscopy, will be necessary for providing definitive answers to some of these questions.
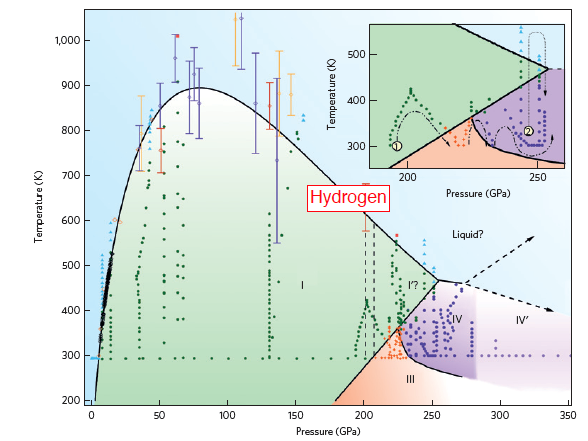
Caption: Phase diagram of hydrogen. The inset shows the P–T paths taken during temperature cycles. The properties of the newly discovered molecular phase, and the question of what happens in the region at higher pressures and temperatures (shown as shaded area), represent exciting areas for future research. Some possible extensions of the phase boundaries are shown by dashed arrows.
Dr. Wendy L. Mao wrote a article to introduce this finding. The article is as following:
![]() Compressed hydrogen heats up - The finding of a new molecular phase in hydrogen under high pressure and moderate temperature adds to the complexity of its phase diagram.
Compressed hydrogen heats up - The finding of a new molecular phase in hydrogen under high pressure and moderate temperature adds to the complexity of its phase diagram.
