The first nickel polyhydride Ni2H3 - Dr. Jack Binns
OCTOBER 11, 2018
Combining high pressures and temperatures can lead to the formation of unusual new compounds. A new study by a group of HPSTAR scientists applies this method to the search for new transition metal hydrides, finding the first nickel polyhydride Ni2H3. This study was published as a Rapid Communication in Physical Review B.
The search for new high-temperature superconductors and hydrogen-storage materials has driven extensive investigations into metal-hydride systems at high pressures. Theoretical studies have been performed for the majority of the elements in the periodic table and most recently a number of polyhydrides have been formed by the first-row transition metals Co, Fe, and Cr.
At ambient pressures nickel has long been known to form a solid solution containing a low concentration of hydrogen, NiH0.01. This concentration can be increased to NiH by applying pressures above 1.25 GPa, however prior to this study nothing was known above this pressure.
“The solubility of hydrogen in many transition metals increases at higher pressures making polyhydrides more favourable,” said Dr. Jack Binns, the lead author of the study. “However, pressure is often not enough, high temperatures are needed to drive the reaction to completion.”
 Here a group of HPSTAR scientists including Jack Binns, Mary-Ellen Donnelly, Mengnan Wang, Eugene Gregoryanz, Philip Dalladay-Simpson and Ross T. Howie use a combination of high pressures and temperatures to create a new nickel polyhydride, Ni2H3. Supporting theoretical work was performed by Andreas Hermann of The University of Edinburgh.
Here a group of HPSTAR scientists including Jack Binns, Mary-Ellen Donnelly, Mengnan Wang, Eugene Gregoryanz, Philip Dalladay-Simpson and Ross T. Howie use a combination of high pressures and temperatures to create a new nickel polyhydride, Ni2H3. Supporting theoretical work was performed by Andreas Hermann of The University of Edinburgh.
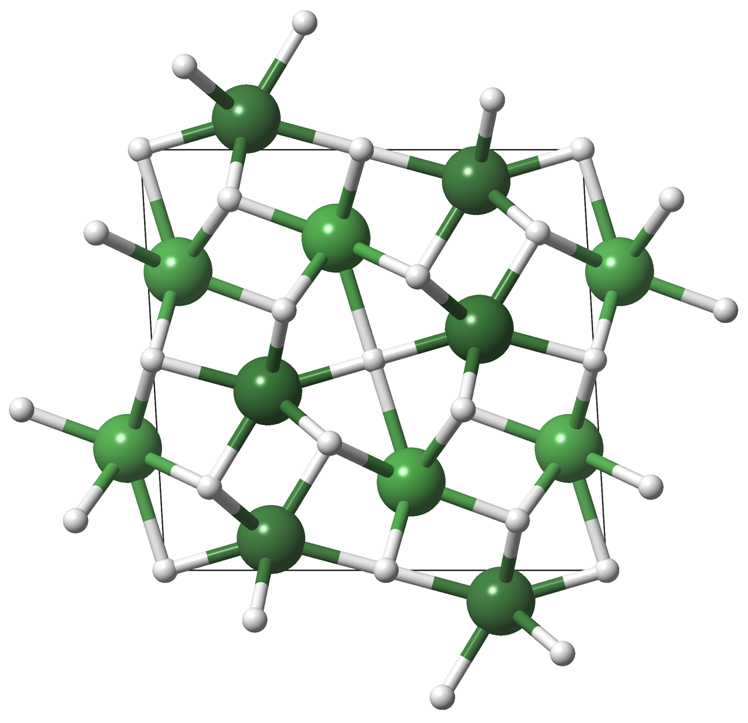
The team found that laser heating a sample of NiH above 1000 K at 50 GPa initiated a transition to a phase crystallizing in a complex new structure. A combination of X-ray diffraction and DFT calculations confirmed the stoichiometry, Ni2H3, and located the hydrogen atoms which are normally invisible to X-rays. They found that this new material transformed back to NiH below 25 GPa and interestingly this phase remains stable to atmospheric pressures and slowly releases molecular hydrogen gas.
“Our study shows that there are still many new phases to discover in metal-hydrogen systems”, said Dr. Ross Howie, the corresponding author “More hydrogen-rich compounds could be formed at even higher pressures.”
Unusually, Ni2H3 contains hydrogen atoms bonded to just four nickel atoms in a flat square, isolated atoms like this are thought to be the first step in the formation of atomic hydrogen layers which may display some of the long sought after properties of metallic hydrogen.
Caption: The complex structure of Ni2H3 containing isolated H atoms (white), surrounded by squares of Ni (green).
硫-氢体系中记录性的超导转变温度的发现,使得富氢化合物的研究成为了科学家们研究的热点课题。理论及实验指出富氢化合物由于非氢元素对氢会产生预压作用,而相对更容易实现金属化,及室温超导。北京高压科学研究中心的Jack Binns博士后带领的研究小组使用原位高温高压实验技术,观测到了镍-氢体系可能存在的化合物。他们发现在大约1 GPa,镍与氢气会形成NiH,NiH可以稳定保持到52 GPa。52 GPa以上,该研究小组对NiH进行激光加热,发现NiH会与氢气进一步生成面心立方结构的Ni2H3。Ni2H3在泄压到25 GPa左右又会转变成NiH与氢,直到常压下,NiH会慢慢的释放氢。
